Nanochemistry and microsctructure of human dental tissues
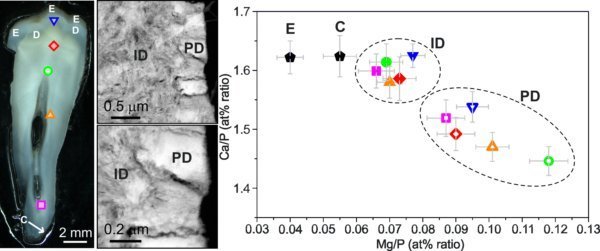
Human mineralized tissues are composed of the inorganic Ca phosphate component hydroxylapatite (HA) and an organic component, mainly collagen. Structurally and chemically very complex biological HA could be described as Ca-deficient carbonate-containing apatite modified mainly with sodium, magnesium, potassium and zinc. Human teeth are composed of three unique dental hard tissues – enamel (E), dentine (D), and cementum (C). Numerous dentinal tubules (DT) penetrate the dentine (D); they are surrounded by a wall of highly mineralized peritubular dentine (PD) and are separated by intertubular dentine (ID).
Our quantitative EDX measurements show constant Ca/P at% ratios across the DEJ; no additional elements could be detected at the junction. A nearly constant Ca/P at% ratio was detected in enamel, ID and cementum with only minor variations in the Mg/P at% ratios. In contrast, a significant lower Ca/P at% ratio was measured in PD compared to ID which is closely related with higher and variable Mg/P at% ratios. Our results suggest that magnesium is most likely incorporated in HA lattice by substituting calcium. Finally, it is important to stress the improvement of TEM specimen preparation used for our study. Applying L-N2 cooling during the ion-milling process seems to be mandatory for avoiding the introduction of artifacts during the preparation of mineralized biological materials.
Outstanding microstructure-controlled properties are the reason for growing interest in natural organic-inorganic materials. Such materials have been an inspiration for producing man-made functional composites. The complexity of composite materials, where inorganic and organic components co-exist, demands structural and chemical characterization performed at high spatial and energy resolution. Therefore, (S)TEM with its variety of imaging and analytical techniques is an advanced approach for characterizing biological composite materials.