Electrocrystallisation
High oxidation states in oxides are not easily achieved by high temperature techniques even under elevated oxygen partial pressures. An elegant and powerful alternative is the anodic oxidation of suitable electrolytes. The electrode plays the role of an oxidising agent with tunable redoxpotential.
|
|
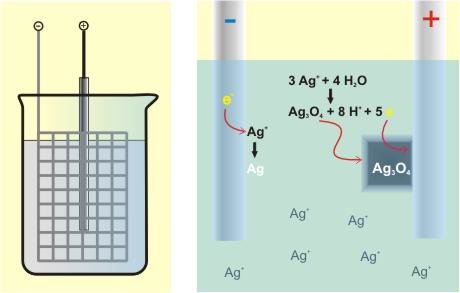
For a specific set of parameters it is possible to oxidise ions in solutions or melts and crystallise solids at the electrode. This happens in liquid phase and so diffusion is directly coupled to current. As a consequence reaction temperatures can often be drastically reduced. The phase separation of electrode and electrolyte allows very pure substances to be synthesised. The direct control of material transport to the electrode, e.g. by a constant current, offers the possibility to optimise the crystal quality of the material. By electrocrystallisation also metastable phases are accessible.
Examples:
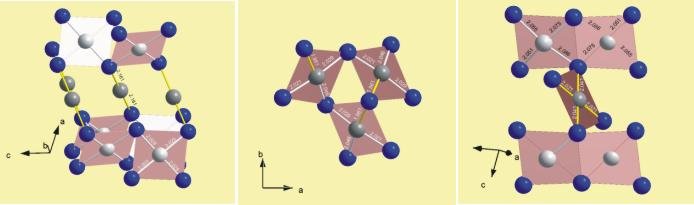
The advantages of electrocrystallisation are opposited by a huge set of possible parameters for optimisation. Besides current density and deposition potential, kind and redox stability of the electrolyte, solubility of the ion to be oxidised, the electrode material, temperature and conductivity of the formed product play an essential role. Impressing examples for the power of this method are the higher silver oxides AgO, Ag2O3 and Ag3O4, in which silver is obtained in the uncommon oxidation state +2. By slight variations of reaction conditions crystals of each substance could be grown and it was possible to determine the structures of these compounds for the first time.
|
|
|
Left: AgO Center: Ag2O3 Right: Ag3O4 |
PrO2 and TbO2-x, which are also accessible by high temperature and high pressure techniques, can be obtained by electrolysis of Pr or Tb containing alkalihydroxide melts at ambient pressure. For TbO2-x a variation of the reaction conditions appeared to have an influence on the resulting TbIII/TbIV ratio and gives rise to discrete defect structures.
|
|
|
TbO2-x- Growth on anode – SEM- picture – XRD- pattern of samples from electrolytes of different temperatures |
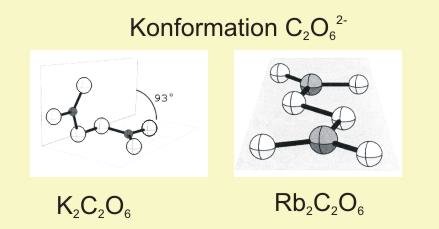
Not in all cases it is possible to obtain single crystals for structure determination. Examples are K2C2O6 and Rb2C2O6 for which a steady growth at the electrode is prevented by their low electrical conductivity. The synthesis of these compounds as a microcrystalline powder was possible by electrolysis of a solution of the respective carbonate. This lead to the first analysis of constitution and conformation of the peroxodicarbonate anion by means of refinement of synchrotron powder diffraction data.
|
|
Publications:
- B. Standke, M. Jansen: Ag2O3, ein neues binäres Silberoxid
Angew. Chem. 97 (1985) 114-115; Angew. Chem. Int. Ed. Engl. 24 (1985) 118-119 - B. Standke, M. Jansen: Ag3O4, das erste Silber(II, III)- oxid
Angew. Chem. 98 (1986) 76-90; Angew. Chem. Int. Ed. Engl. 25 (1986) 77-78 - M. Jansen, P. Fischer: Eine neue Darstellungsmethode für monoklines Silber(I, III)- oxid (AgO). Einkristallzüchtung und Röntgenstrukturanalyse
J. Less-Common Met. 137 (1988) 123-131 - U. Bilow, M. Jansen: Electrocrystallization and crystal structure determination of Ph4PC60 * Ph4PC1
J. Chem. Soc. Chem. Commun. 1994 (1994) 403-404 - M. Malchus, M. Jansen: Electrocrystallization of PrO2 and TbO2-x from alkali hydroxides melts and characterization of the fluorite- related TbO2-x
Solid State Sci. 2 (2000) 65-70 - M. Jansen, S. Vensky: Darstellung und Charakterisierung des Silber(I, II, III)- oxidclathrates Ag7O8HCO3
Z. Naturforsch. 55 b (2000) 882-886 - R. E. Dinnebier, S. Vensky, P. W. Stephens: Crystal structure of K2[C2O6] - First proof of existence and constitution of a peroxodicarbonate ion
Angew. Chem. 114 (2002) 2002-2004; Angew. Chem. Int. Ed. Engl. 41 (2002) 1922-1924 - S. Vensky, L. Kienle, R. E. Dinnebier, M. Jansen: Zur Realstruktur von Na3BiO4
Z. Kristallogr. Suppl. 21 (2004) 186 - R. E. Dinnebier, S. Vensky, M. Jansen: Crystal and molecular structure of rubidium peroxodicarbonate Rb2[C2O6]
Chemistry – A European Journal 9 (2003) 4391-4395