Intercalation and Deintercalation
The interplay between structure, electronic state and properties of solid state compounds is studied best at systems with modifiable composition. Upon extracting particles out of a solid the host is simultaneously oxidised. This forces a well defined change in the valence electron concentration which is directly coupled to the properties. In the focus are coinage metals, which are ideal guest species, while host materials can vary in composition and dimensionality of the Van-der-Waals gap.
|
|
As an experimental tool, electrochemical techniques are the method of choice, because they allow selective insertion / extraction of the mobile species and a topotactic control of reaction. The degree of de- / intercalation is precisley controlled by monitoring the charge and the measured cell voltage gives hints on thermodynamic and kinetic features of the investigated system. By this way mixed valent compounds can be synthesised and changes in their properties can be observed.
The choice of different cell setups makes it possible to accomplish de- / intercalation reactions for different types of ions and at different temperatures. The use of multipotentiostats allows potentiostatic and galvanostatic processing.
Example: Electrochemical Intercalation
Concerning their structures and electronical properties, many chalcogenides of the transition metals are suitable as host materials for the intercalation of guest species. If one is restricted to cationic guests, the appropriate amount of electrons has to be transferred simultaneously to the host during intercalation, to preserve electroneutrality. This process can be accomplished either electrochemically by the use of multipotentiostats and diverse cell types or "conventionally" by the utilisation of capable reduction reagents. From a thermodynamic point of view, the corresponding metal is suitable as reduction reagent, as intercalation of M+/e- is only successful, if a driving force is present, reflected by the relationship μMetall(M+)>μWirt(M+). For a reversible intercalation into the host compound, the structurally essential Van-der-Waals gaps must be preserved upon intercalation. This means, that from a structural point of view, the reaction must be of topochemical character, so, in the ideal case, the elements of the lattice array are preserved concerning their mutual orientation. Apart from this, as an electronic prerequisite, the host compound must show low lying, and therefore easily accessible bands right above the Fermi level, which is fulfilled for metallic conductors and semiconductors with small band gaps. The guest species preferably are singly charged cations from the 1st main or 1st transition group. Regarding the alkaline metals, lithium is in a prominent position. According to its very small ionic radius and its highly negative standard reduction potential, it is an outstanding candidate for the guest species. Its low molecular mass is another reason for the most intensive investigation of its intercalation behaviour, especially as application in primary and secondary battery systems.
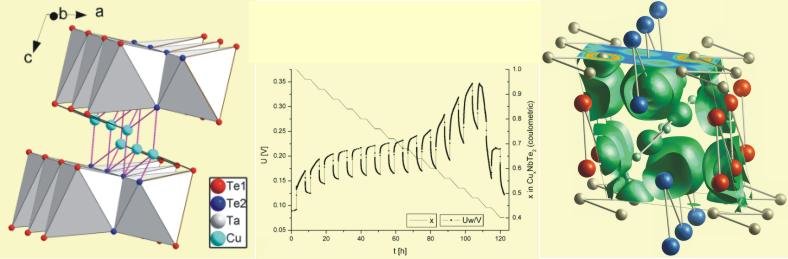
We are interested in the intercalation chemistry of coinage metals, especially of copper and silver. The single positive charged ions, in contrast to those of the alkaline metals, show no noble gas configuration, but a d10- configuration. Interesting effects are related to this configuration. The higher the atomic number, the more pronounced are the so-called attractive d10-d10-interactions. This is amongst others responsible for the formation of separated cationic structures in many solid state silver compounds. For copper, this behaviour is also expected, but it is supposed to be much weaker than with silver. Concerning the intercalation behaviour, copper has the advantages of a smaller ionic radius and a less positive standard reduction potential over silver. From the transition metal dichalcogenides with a two dimensional Van-der-Waals gap, the ditellurides of the 5th transition group were broadly investigated. Copper intercalates between the MTe2 sheets, which are shifted against each other, so that copper is coordinated tetrahedrally by tellurium. The so formed tetrahedral sites can almost be fully occupied by copper.
|
|
|
Left: crystal structure of Cu0,89TaTe2 Center: cell potential during coulometric titration Right: Plot of the electron localisation function (ELF) |
The maximum occupancy was achieved by "conventional" reduction of the host compound with the element (here Cu/CuCl in acetonitrile/MTe2) at elevated temperatures in a closed system (autoclave). Different compositions are adjusted electrochemically by the use of the MacPileII-System by coulometric titration. The experiments are conducted with constant cell current during charge (Galvanostatic Intermittent Titration Technique). The drop in cell voltage (during charge and relaxation) is characteristic for the decomposition of the sample (shift from one to two phase mechanism). Prior to this voltage drop, the samples are single phase and can be removed from the cell for further investigations. Furthermore, the performed band structure calculations (TB- LMTO-ASA) and the analysis of the Electron Localisation Function (ELF) allow a deeper insight in the bonding situations.
Example: Electrochemical Deintercalation
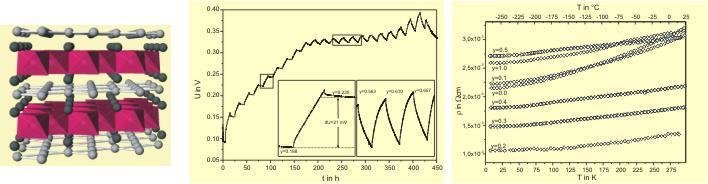
A representative example for a (reversible) deintercalation of silver out of a ternary oxide is Ag5Pb2O6. Due to structural features and the electronic conductivity, this compound can be considered as Ag+5Pb24+O6(e-). The homogeneity range (x = 5.02(1) - 4.51(1)) of AgxPb2O6 was determined by chemical analysis and Rietveld refinement of powder diffraction data. The obtained structural data shows that those Ag ions, which are placed between the PbO3 sheets, are topotactically removed. For higher degrees of deintercalation (0.5 < y < 1.0) the cell voltage remains constant, indicating an equilibrium between a solid solution and new phase Ag4Pb2O6. The electrical conductivity is influenced by a change in the valence electron concentration.
|
|
|
Ag5Pb2O6- crystalstructure – changes in cellpotential during coulometric titration – temperaturedependence of the specific electrical resistance of samples with a different degree of deintercalation. |
Publications:
- K. E. Adelsberger, J. Curda, S. Vensky, M. Jansen: High-pressure synthesis and electrochemical investigation of Ag2Cu2O3
J. Solid State Chem. 158 (2001) 82-86 - K. E. Adelsberger, L. Prigent-Croguennec, M. Jansen: Electrochemical deintercalation of Ag2PbO2
Z. Anorg. Allg. Chem. 627 (2001) 2473-2476 - T. Sörgel, M. Jansen: Structure refinement, physical properties and electronic structure of new electrochemically copper intercalated group Vb ditellurides CuxMTe2 (M = V, Nb, Ta)
Solid State Sci. 6 (2004) 1259-1267 - C. P. M. Oberndorfer, K. E. Adelsberger, M.Jansen: Elektrochemische Deinterkalation von Ag5Pb2O6
Z. Anorg. Allg. Chem. 631 (2005) 385-392