
Inorganic Ozonides
Inorganic ozonides are compounds containing the radical anion O3–, a 19-valence- electron anion, showing a conspicuous red color. Due to extreme air-, moisture- and temperature sensitivity, gram amounts of pure alkali ozonides have been synthesized not until 1985, starting from alkali hyperoxides and ozone.[1] This procedure permits the synthesis of the higher alkali ozonides (K – Cs), the starting materials of any further ozonide chemistry.[2]
All other ozonide compounds require other/new synthesis routes e.g. metathesis reactions[3] or ion exchange.[4]
| An ozonide crystal sealed in a capillary.[3] | 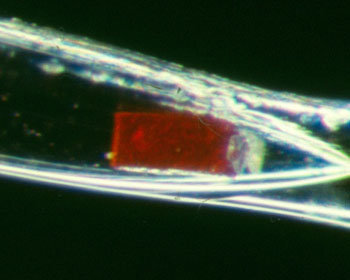 |
Meanwhile, the field of ozonides has been expanded to:
|
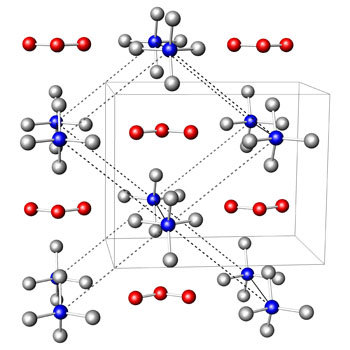 |
Recent results:
- Cs5([12]crown-4)2(O3)5 - the first supramolecular ozonide compound[11]
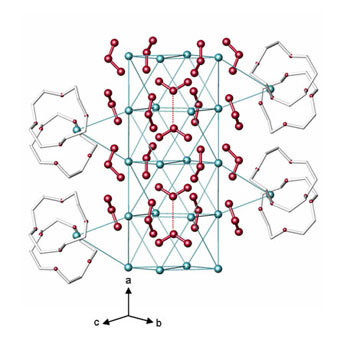 |
The reaction of [12]crown-4 with CsO3 in liquid ammonia yields this compound. Left: The prominent structural feature are 1∞{Cs8(O3)10}2- rods which are arranged in a tetragonal rod packing (see below). |
| The structural confinement enforced by the small crown ether molecules lead to remarkably short intermolecular O⋅⋅⋅O distances indicated by the dotted red line in the figure above. | 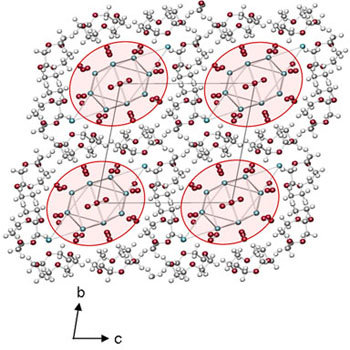 |
A recent review:
- M. Jansen, H. Nuss: Z. Anorg. Allg. Chem. 2007, 633, 1307.
References:
- W. Schnick, M. Jansen:Angew. Chem. Int. Ed. 1985, 24, 54.
- W. Hesse, M. Jansen, W. Schnick: Prog. Solid St. Chem. 1989, 19, 47.
- W. Hesse, M. Jansen: Inorg. Chem. 1991, 30, 4380.
- N. Korber, M. Jansen: Chem. Ber. 1992, 125, 1383.
- W.Klein, M. Jansen: Z. Anorg. Allg. Chem. 2000, 626, 947.
- W. Klein, M. Jansen: Z. Naturforsch. 2005, 60b, 426.
- W. Assenmacher, M. Jansen: Z. Anorg. Allg. Chem. 1995, 621, 431.
- H. Seyeda, K. Armbruster, M. Jansen: Chem. Ber. 1996, 129, 997.
- P. D. C. Dietzel, M. Jansen: Z. Anorg. Allg.Chem. 2006, 632, 2276.
- N. Korber, M. Jansen: J. Chem. Soc., Chem. Commun. 1990, 1654.
- H. Nuss, M. Jansen: Angew. Chem. Int. Ed. 2006,45, 7969.
- H. Nuss, J. Nuss, M. Jansen: Z. Anorg. Allg. Chem. 2008, 634, 1291.
- H. Nuss, M. Jansen: Z. Naturforsch. 2009, 64b, 1325.