Nitrides
Synthesis of Na3N
One of the most pronounced violations of the homology principle in the periodic table is found in the binary nitrides of the alkali metals. While Li3N forms spontaneously from the elements at room temperature, the synthesis of the homologues Na3N and K3N has failed so far in spite of a multitude of attempts during many decades. The synthesis of Na3N succeeded via simultaneous deposition of sodium with activated nitrogen onto a sapphire substrate in the temperature range between 77 and 300 K. Na3N crystallizes in the anti-ReO3 structure type and decomposes already at 360 K to the elements.
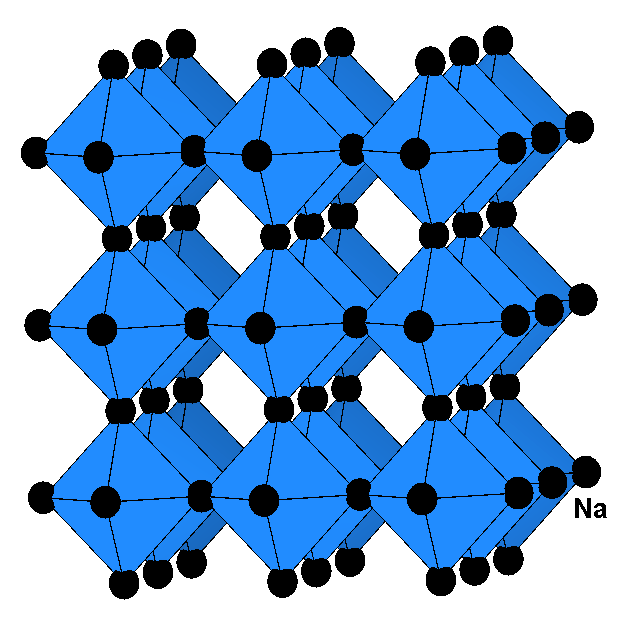 |
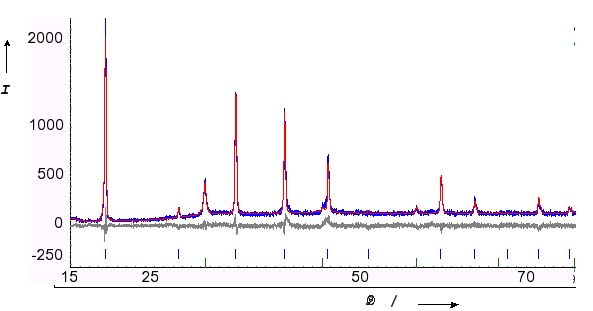
Structure type: anti-ReO3 cubic, space group number 221 (PM-3M) Z=1, Na: (3d) N: (1a) a = 473.2 pm d(Na-N) = 236.6 pm Rp = 8.0 %, Rwp = 10.3 % |
|
|
|
Rietveld refinement: blue (measurement), red (calculated), grey (difference) |
Reference: D.Fischer, M. Jansen, Angew. Chem. 114 (2002) 1831-3; Angew. Chem. Int. Ed. 41 (2002) 1755-6.
Synthesis of K3N
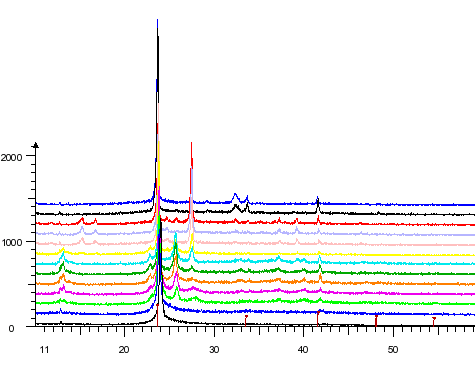
Two phases in the binary system K/N have been obtained via co-deposition of potassium and nitrogen at 77 K (subtrate temperature) and subsequent heating. The powder pattern of one of these phases can satisfactorily interpreted by assuming the composition K3N and the anti-TiI3 structure type, which is also adopted by Cs3O.
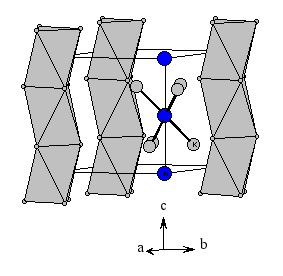 |
Structure type: anti-TiI3 193 K, hexagonal, space group number 193 (P63/MCM) Z=2, K: (6g) N: (2b) a = 780 pm, c = 759 pm d(K-N) = 278 pm |
|
|
|
Temperature dependent X-ray powder pattern of a K/N co-deposition. |
Reference: D.Fischer, Z.Cancarevic, J.C. Schön, M. Jansen, Z. Anorg. Allg. Chem. 630 (2004), 156-160.