Alloys
CaLi2
Laves phases represent the largest group of intermetallic compounds and are found to crystallize in three structure types (MgCu2, MgZn2, MgNi2). Various concepts (including geometrical and electronic approaches) have been utilized in order to predict the occurrence of Laves phases in real systems. However, in practice this conceptual approach is of limited value. Therefore new studies of the phase relations and stability ranges of Laves phases are being carried out under the inter-institutional research initiative entitled “The Nature of Laves Phases” http://laves.mpie.de.
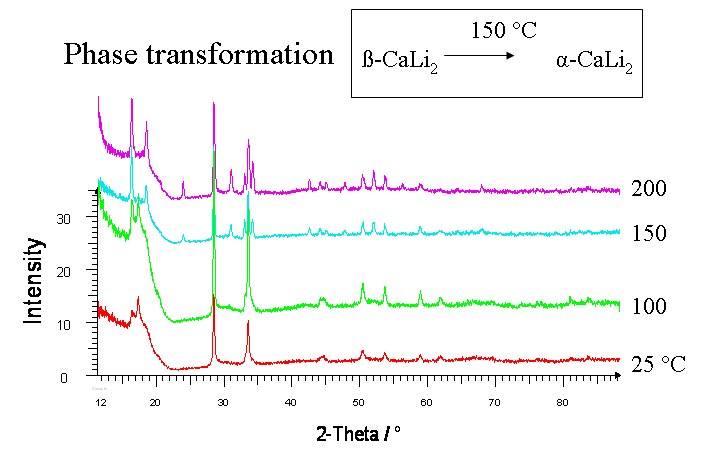
A new modification of CaLi2 (β-CaLi2) was obtained by simultaneous deposition of calcium and lithium on a sapphire substrate. This compound crystallizes in the cubic MgCu2-structure type (Fd-3m, a = 885.6(1) pm, Rietveld-method) and transforms at 150°C into the since 1943 well known hexagonal α-CaLi2.
|
|
|
X-ray powder patterns of CaLi2; 25°C (β-CaLi2), 150°C (α-CaLi2). |
Reference: D.Fischer, M. Jansen: Z. Anorg. Allg. Chem. 629 (2003) 1934-6.
LiZn13
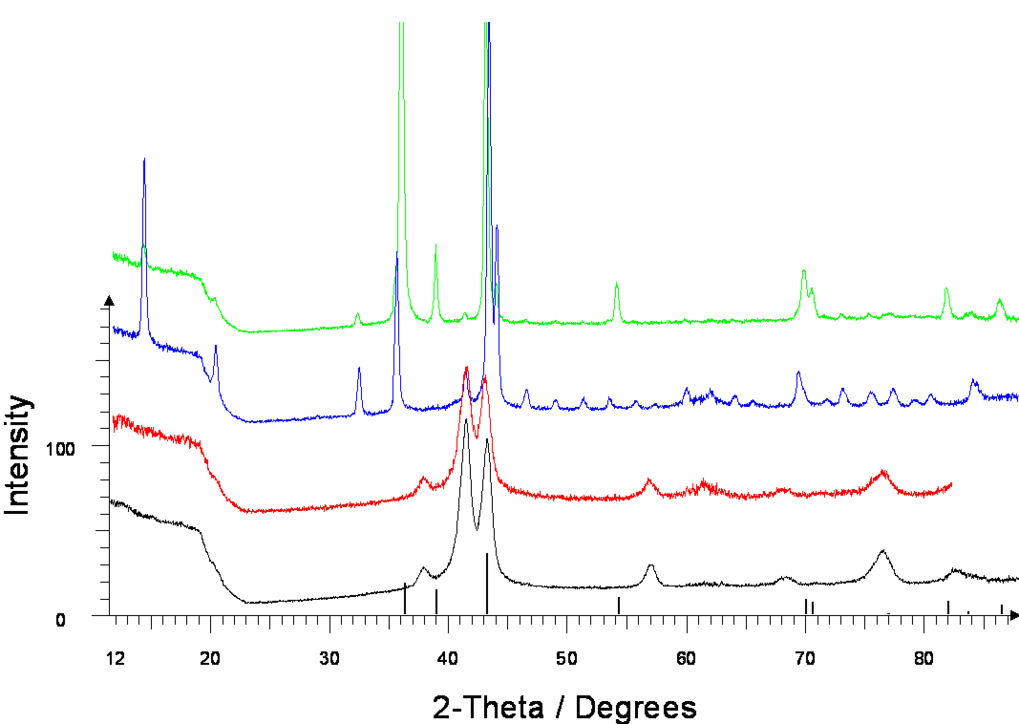
The cubic NaZn13 type of structure is genuinely adopted by binary alloys of the alkali metals Na, K, and Rb with zinc. But for the Li-Zn system this compound is unknown. Co-depositing zinc and lithium with a ratio of Zn:Li = 13:1 onto sapphire substrates at –196°C results in grey-metallic samples, which show diffraction patterns of a hexagonal-closed-packed phase (β’-phase) up to –50°C. By subsequent warming up to room temperature, the samples transform completely into the new phase LiZn13. The structure determined by the Rietveld method reveals LiZn13 to be isostructural to NaZn13 (Fm-3c, a = 1234.83(6) pm). Furthermore a slow transformation of LiZn13 into zinc occurs already at room temperature.
|
|
|
X-ray powder patterns of Zn/Li-depositions at -196°C (Zn:Li=13:1); measured at: -19°C: β’-phase, -50°C: β’-phase, 25°C: LiZn13, 160°C: Zn from bottom to top; tick marks Zn. |
Reference: D.Fischer, M. Jansen: Z. Anorg. Allg. Chem. 636 (2010) 1917-9.