Atoms and molecules on the same wavelength
Organic dye molecules are used as single photon sources and can be tuned to the optical transitions of alkali metals
It may be surprising, but in physics the terrain of atoms and the territory of organic molecules are worlds apart. Therefore, in order to have a molecule communicate optically with atoms, the physicists must put in a similar amount of effort as most Europeans do when they want to learn Chinese. A team from the Max Planck Institute for Solid State Research and the University of Stuttgart has now initiated a communication between molecules and atoms for the first time. The researchers have forced the molecules to emit a stream of single photons. They selected the molecules such that the colour of their emission was perfectly tuned to the absorption of sodium atoms. Subsequently, the researchers were able to develop a filter from a glass cell filled with sodium vapour that blocks light of this colour extremely well and could be interesting for biological experiments, for example. In addition, the physicists used the gas-filled cell to slow down the single photons and store the photons temporarily for a few nanoseconds. This could, for instance, be of interest for quantum communication which pursues completely new approaches in the transmission and processing of data.

Ilja Gerhardt and his colleagues are building bridges in physics: The researchers from the Max Planck Institute for Solid State Research in Stuttgart and the university there have brought together atomic and molecular physics in their experiments. To the layperson, the two disciplines may be closely related; but for physicists, their territories are separated by an as yet rather impenetrable border. One of the reasons why no one has so far made molecules talk to atoms is because the interchange between scientists from the two fields is not very frequent either.
Moreover, the experiments with molecules have a very different set of requirements to those of experiments with atoms, and offer other opportunities. “We have now combined the spectroscopy of individual molecules with the spectroscopy of atoms for the first time,” says Ilja Gerhardt, who headed the experiment. Spectroscopy is the workhorse of the quantum physicists. The researchers irradiate a material with light – usually from a laser – and observe whether light is emitted by the substance and, if so, which light. This tells them a lot about the structure and the behaviour of their test objects.
A particularly bright source for single photons
The researchers have now made a single molecule emit single photons which can be absorbed by atoms. Nowadays, there are a number of sources of single photons; they are characterised by the fact that they never emit two or more photons simultaneously. Single photons are of interest as carriers of quantum information as they can be transmitted through fibre optics, for example, and their quantum state cannot be copied. They could, thanks to a method called quantum cryptography, make it possible to have interception-proof communication. “Our source of single photons is many times brighter than others,” says Ilja Gerhardt. This is down to the fact that the molecules used by the Stuttgart-based researchers emit more than half a million photons per second, and thus 1000-times more photons than other sources, but in a continuous stream of single photons. Particularly bright sources of single photons could significantly accelerate the transmission of quantum information because they can transmit more information per second.
The molecules shine, however, not only very brightly, but also in a special light. “We’ve caused a molecule to talk with the wavelength of an atom,” says Ilja Gerhardt. The first task was to select the right molecules and atoms. Ilja Gerhardt scanned the literature to learn which atoms and molecules absorb and emit light of approximately the same wavelength – i.e. can be combined well. This depends mainly on which energy states the electrons of the candidates can assume. When an electron jumps from one energy level to a higher one, it absorbs a photon in the process, as it requires its dose of energy. If it falls from one level to a lower one, it may emit a photon. This is also the case with individual organic molecules. Usually, the molecules emit red-shifted photons, i.e. photons with less energy – similar to a fluorescent marker which fluoresces orange if it is irradiated with blue light.
Molecules which speak the optical language of atoms
Ilja Gerhardt identified the aromatic compound dibenzanthanthrene and sodium as one suitable pair. The reason is that the dibenzanthanthrene molecules speak the optical language of the atoms: they emit an orange-yellow light that can be absorbed by sodium atoms.
Not all molecules emit light of exactly the same colour, however, each talks in its own dialect, so to speak. In order to identify one which the sodium can understand as well as possible, the researchers initially prepared a solution of the molecules. They diluted it so much that the molecules had a lot of space for themselves. They then froze a thin film of the solution at 1.4 degrees above absolute zero - minus 273.15 degrees Celsius. In this freezing cold, the molecules hardly move so that they absorb and emit light of sharply defined wavelengths.
The researchers now irradiated the sample through a microscope with a laser beam of the colour which sodium atoms also absorb. They then observed the molecules, which get excited by the orange-yellow light, under the microscope. The only molecules excited were those that could also communicate with this light. The energy loss between absorption and emission causes the molecules to show up in red light.
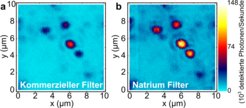
The red flashes of the molecules are drowned by the light of the exciting laser, just as the stars in the night sky can no longer be recognised from a floodlit stadium. The researchers therefore filter out the exciting light from the beam which is directed to the detector. A simple red glass would actually be sufficient to see an individual molecule; however, the glass unfortunately also filters out a portion of the red light and therefore acts like sunglasses – a vast amount of photons are thereby lost.
A sodium vapour filter could make biological experiments easier
“We discovered that a cell with hot sodium vapour was much more suitable for this than commercial filters,” says Ilja Gerhardt. This is because the sodium vapour at a temperature of 155 degrees very precisely removes only the orange-yellow light from the beam, while commercial filters always remove some light from other colours as well. The light spots of the molecules are therefore much easier to see through the sodium vapour filter than through a commercial one. “Our discovery is very relevant for biological experiments, for example,” says Ilja Gerhardt. Biologists often investigate the processes in cells by equipping proteins with fluorescent labels and excite them with laser radiation. Their investigations could provide much more information with a more precise filter, as they make it easier for them to capture the signals they are interested in.
This practical finding for experimenters who work with lasers is based on only one of the possible variants in the optical interplay between molecules and atoms, however. With this experiment it is not yet possible for molecules and atoms to communicate with each other because the sodium atoms are blind, or, to stay with the same analogy, deaf to the red light of the molecules. But if molecules can absorb orange-yellow light which is understood by sodium atoms, they can also emit it – the researchers have only to slightly modify their experiment. To this end, they first selected one of the molecules from the deep-frozen solvent that talks the photonic language of the sodium.
A reversed sodium filter identifies the colour of the molecular flashes
In order to now filter out the light which coincides precisely with the sodium line, the researchers in Stuttgart used a trick: the sodium vapour cell is again helpful here, because its filter effect can be reversed by a magnetic field. It then allows only light to pass with an orange-yellow colour, usually absorbed by the sodium atoms. The researchers therefore sent the flashes of a molecule through the reversed sodium vapour cell onto a single photon counter. If this was now triggered, they could be sure that the molecule had spoken in the language of the sodium.
“In the molecule we’ve therefore found a brilliant single-photon source whose light we can tune to the absorption of atoms,” says Ilja Gerhardt. This could be helpful for processing of quantum information, as the information carried by scurrying photons can thus be recorded in atoms.
Sodium vapour as a temporary memory for single photons
Lastly, the Stuttgart-based researchers also proved that the sodium vapour cell can store the light of a molecule temporarily. This means that the cell perceptibly brakes the flow of single photons. The researchers observed this when they made a molecule emit orange-yellow light which was very close to the colour of the sodium light, but differed slightly from it. Therefore, it was just able to pass the sodium vapour cell, but could be still slowed down. With an ingenious stopwatch for photons, the scientists proved that a photon exits the sodium vapour cell only after a delay of six nanoseconds.
The temporary memory for photons will be useful for processing quantum information with a quantum computer, for example, in a very similar way to the temporary memories of today’s electronics. Before this point is reached, the physicists still have to come to grips with many fundamental and practical problems, however. The communication between molecules and atoms must also improve. Be this as it may: the dialogue has now begun.
PH

