Intermetallic Phases
Jürgen Nuss, Ulrich Wedig, Sabine Prill-Diemer, Claus Mühle, Fei Wang
Among the classes of inorganic solids, the intermetallics are the ones that are least understood with respect to their bonding properties. Unlike ionically or covalently bonded compounds, it is virtually impossible to apply simple heuristic concepts to reliably deduce compositions or structural features from the elements involved and from the valence electron concentration per atom (VEC) provided. Also, the simple Fermi liquid picture usually fails to properly explain even basic physical properties such as electrical conductivity and magnetic susceptibility. This is mainly due to the fact that some of the features that primarily determine the chemical bond, such as the degrees of valence electron transfer and localization, vary strongly and almost continuously with composition and nature of the elements involved.
Platinides
Introduction
The importance of relativistic effects grows with increasing atomic number and becomes rather prominent when reaching the 6th period. The effects are particularly noticeable for gold, which exhibits the highest electron affinity among all metals (2.31 eV). The auride anion Au– is stable in the gas phase, in solution and in the solid state. Platinum is the metal with the second highest electron affinity (2.13 eV) after gold, exceeding that of any chalcogen (highest value 2.08 eV; sulphur) which readily form, as typical non-metals, negatively charged dianions. Due to this fact it appeared promising to attempt the synthesis of platinides containing platinum with a twofold negative ionic charge realizing the closed-shell valence state.
Cs2Pt
|
 |
BaPt
|
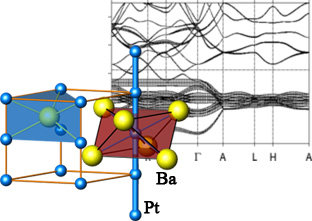 |
Ba2Pt
|
 |
Geometric variations and electron localizations in PbFCl type compounds
Introduction
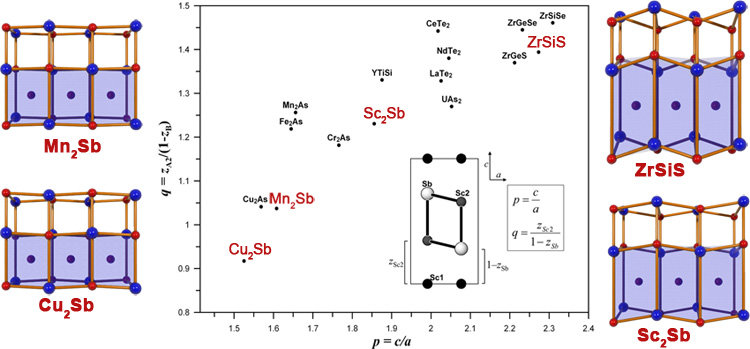
The geometric structures of intermetallic phases are determined by a complex interplay of various bonding types. Ionic and covalent interactions caused by charge transfer among the atoms and by local electronic structures compete with the forces that itinerant electrons exert on the atomic cores. Compounds with the general formula A2B, crystallizing in the anti-PbFCl type of structure, were investigated exemplarily by experimental and theoretical means to demonstrate the complexity of chemical bonding in intermetallic phases. The PbFCl structure is adopted by about 540 known compounds. All of them are isopointal, i.e. they crystallize in the same space group (P4/nmm) and the atoms occupy the same sites c2a (tP6): F in 2a, Cl in 2c and Pb in 2c. In the A2B intermetallic compounds regarded here, a transition metal A takes up the positions of F (A1) and Cl (A2) and a pnictogen atom B (As or Sb) that of Pb.
Results and Discussion
The variations in the structural details are reflected by different c/a ratios and the spread of the two free atomic parameters zA2 and zB. The ratio of the latter ones q = zA2/(1–zB) is a measure of the unevenness of the (A2)B layer and may be plotted against p = c/a in order to generate a structure field diagram (fig.). Compounds with q larger than 1.5 are generally ionic or very polar. The low q value together with a rather low c/a ratio is characteristic for the intermetallic representatives and corresponds to a moderate or in the case of Cu2Sb even reversed buckling of the (A2)B layers (fig.).
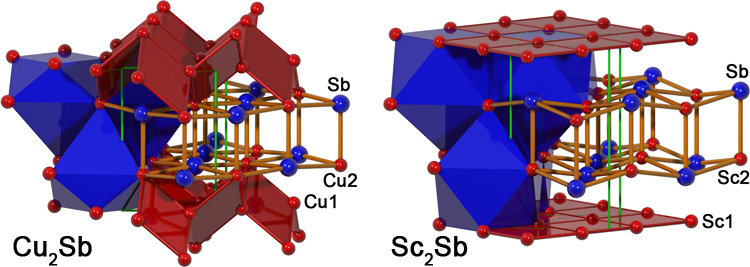
Because of these geometrical features, the description of the structures in terms of coordination polyhedra, which is suitable for the more ionic compounds, is less clear. The intermetallic compounds may be regarded more appropriately as arrangements of stacked layers or as variants of the NaCl structure, where half of the (A2)4B4 heterocubane units are filled by A1. Cu2Sb is unique among the compounds investigated here, being the only representative with q < 1. The Cu1—Cu2 distance (260 pm) is much shorter than d(Cu1—Cu1) = 283 pm, suggesting at first sight Cu—Cu attractive interactions in the nonplanar 44 nets of Cu1 and Cu2, as highlighted by red planes in the left part of the figure. However, d(Cu1—Cu2) is still larger than the atomic distance in elemental copper (256 pm). The relation of the geometrically different A—A distances is inverted when going from Cu2Sb over Mn2Sb to Fe2Sb, and finally increases via Sc2Sb continuously to ZrSiS. This trend is associated with an increase of p and q. The distance between two neighboring Sc1 atoms is 298 pm, 45 pm shorter than d(Sc1—Sc2) and even 24 pm shorter than the atomic distance in metallic scandium. This indicates metal-metal interactions in the planar 44 nets formed by Sc1 only, which are highlighted by a red plane in the right part of the figure. Even stronger interactions between the A1 components (Si) were expected in ZrSiS, according to its position in the structure map.
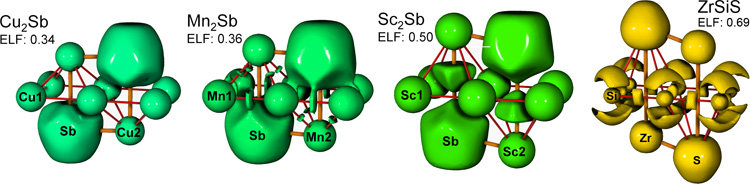
To investigate how local bonding patterns influence the geometric structure, the corresponding scalar fields of the Electron Localization Function (ELF) were analyzed. Selected domains of the ELF are shown in the figure below. Two center Si—Si bonds with a bond order of ½ were deduced from the ELF of ZrSiS. In addition, the charge distribution as obtained from the topological analysis of the ELF and of the electron density justifies the formal description of ZrSiS within the Zintl-Klemm concept as [Zr4+S2–]2+[Si]2–. Due to the strong interaction in the Si (A1) planes, ZrSiS holds one of the extreme positions within the structure map. The other extreme position is occupied by Cu2Sb, where no ELF attractors are found besides the domains that are attributed to the atoms. ELF domains in the valence region between the transition metal atoms appear in Mn2Sb. The same ELF topology is observed in Cr2As, Mn2As and Fe2As. Compounds like Sc2Sb, containing early transition metal elements, exhibit large ELF basins in the pyramidal voids enclosed by the A1 and A2 atoms. The integrated valence electron density within this basin amounts to 1.3 electrons in the case of Sc2Sb. This localization pattern, which also appears in YTiSi and YTiGe, is according to our detailed analysis an intrinsic property of the ELF topology of this family of compounds and is directly related to the bonding interaction within the planar 44 nets, formed by the A1 atoms.
When going from left to right in the first row of transition metals, the d electrons become more localized. For the compounds A2B under discussion, this leads, on one hand, to a continuous change of the charge transfer from the transition metal to the pnictogen atoms which in Cu2Sb is even inverted. On the other hand, rather peculiar bonding conditions occur, which are reflected by the structural differences. We find multicenter bonding among the early transition metals and direct metal-metal interactions in the Cr, Mn and Fe representatives, whereas Cu2Sb may be described as Cu atoms being embedded in metallic antimony.
J. Nuss, U. Wedig, M. Jansen: Geometric Variations and Electron Localizations in Intermetallics: The Case of La2Sb Type Compounds
Z. Anorg. Allg. Chem. 637 (2011) 1975-1981. doi:10.1002/zaac.201100331V. Saltykov, J. Nuss, M. Konuma, M. Jansen: SrAu0.5Pt0.5 and CaAu0.5Pt0.5, analogues to the respective Ba compound, but featuring purely intermetallic behaviour
Solid State Sciences 12 (2010) 1615-1619. doi:10.1016/j.solidstatesciences.2010.07.011J. Nuss, U. Wedig, M. Jansen: Synthesis, Structure and Properties of Scandium Dysprosium Antimonide, ScDySb
Z. Anorg. Allg. Chem. 636 (2010) 913-918. doi:10.1002/zaac.201000095J. Nuss, U. Wedig, A. Kirfel, M. Jansen: The Structural Anomaly of Zinc: Evolution of Lattice Constants and Parameters of Thermal Motion in the Temperature Range of 40 to 500 K
Z. Anorg. Allg. Chem. 636 (2010) 309-313. doi:10.1002/zaac.200900460V. Saltykov, J. Nuss, M. Konuma, M. Jansen: Investigation of the Quasi Binary System BaAu-BaPt
Z. Anorg. Allg. Chem. 635 (2009) 70-75. doi:10.1002/zaac.200800300D. Errandonea, D. Santamaría-Perez, A. Vegas, J. Nuss, M. Jansen, P. Rodríguez-Hernandez, A. Muñoz: Structural stability of Fe5Si3 and Ni2Si studied by high-pressure x-ray diffraction and ab initio total-energy calculations
Phys. Rev. B 77 (2008) 094113-1-12. doi:10.1103/PhysRevB.77.094113J. Nuss, U. Wedig, M. Jansen: Geometric variations and electron localizations in intermetallics: PbFCl type compounds,
Z. Kristallogr. 221 (2006) 554-562. doi:10.1524/zkri.2006.221.5-7.554A. S. Karpov, M. Konuma, M. Jansen: An experimental proof for negative oxidation states of platinum: ESCA-measurements on barium platinides,
Chem. Commun. 8 (2006)838-40. doi:10.1039/b514631cA. S. Karpov, U. Wedig, R. E. Dinnebier, M. Jansen: Dibariumplatinide: (Ba2+)2Pt2-·2e-, and Its Relation to the Alkaline-Earth-Metal Subnitrides,
Angew. Chem. 117 (2005) 780-783. doi:10.1002/ange.200462055; Angew. Chem. Int. Ed. Engl. 44 (2005) 770-773. doi:10.1002/anie.200462055A. Karpov, J. Nuss, U. Wedig, M. Jansen: Covalently Bonded 1∞[Pt]–-Chains in BaPt — Extension of the Zintl-Klemm Concept to Anionic Transition Metals?
J. Am. Chem. Soc. 126 (2004) 14123-14128. doi:10.1021/ja0401186A. Karpov, J. Nuss, U. Wedig, M. Jansen: Cs2Pt, ein Platinid(-II) mit vollständiger Ladungstrennung, Angew. Chem. 115 (2003) 4966-4969 doi:10.1002/ange.200352314 ; Cs2Pt a Platinide(-II) Exhibiting Complete Charge Separation, Angew. Chem. Int. Ed. Engl. 42 (2003) 4818-4821. doi:10.1002/anie.200352314
J. Nuss, M. Jansen: Demarcation of the PbFCl and Cu2Sb Structure Families: Crystal Structure Re-Determinations and Refinements of CuMgSb, Cu2Sb and CuMgAs,
Z. Anorg. Allg. Chem. 628 (2002) 1152-1157. doi:10.1002/1521-3749(200206)628:5<1152::AID-ZAAC1152>3.0.CO;2-1