NMR Spectroscopy of Inorganic Solids
- Physical Cemistry of amorphous materials
- Ionic conductors
- Host guest interactions in zeolite structures
Physical Chemistry of amorphous solids
The Physical Chemistry of amorphous materials such as ceramics and glasses is one of the corner stones of our research. We are developing modern solid state NMR strategies or the characterization of these systems, allowing for an understanding of the microstructural features. The long term goal of these studies is to correlate the macroscopic physical and chemical properties with the microstructural features, thus being able to optimize specific key properties of these materials.
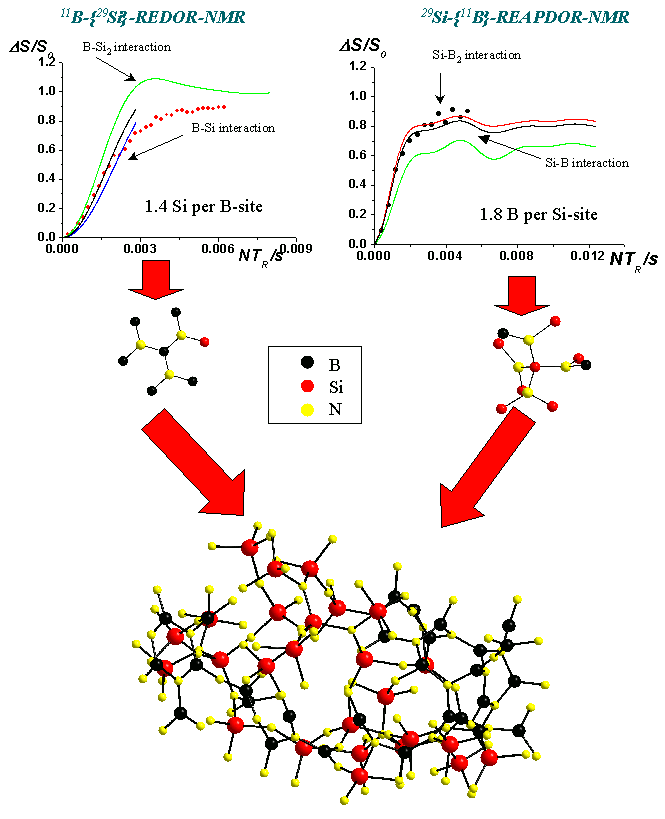
A prominent issue is the arrangement of the network forming polyhedra ((SiO4/2, BO3/2, BO4/2, POO3/2, BN3/3, SiN4/3 ...) into an extended three dimensional network. Our approach is to use the heteronuclear dipolar interaction between two nuclei I and S ( e.g. 11B and 29Si), accessible by modern NMR double resonance approaches such as REDOR, TRAPDOR, TEDOR and REAPDOR. This provides internuclear distances, typically up to 6 Angstrom. From this, we can deduce number and species of the next nearest neighbors.
The example below is taken from a structural characterization of nitridic high performance ceramics. The figure shows 11B-{29Si}-REDOR and 29Si-{11B}-REAPDOR-NMR evolution curves, allowing for a determination of the number of hetero-atoms in the second coordination sphere. From this, network model can be proposed and tested.
Ionic conductors
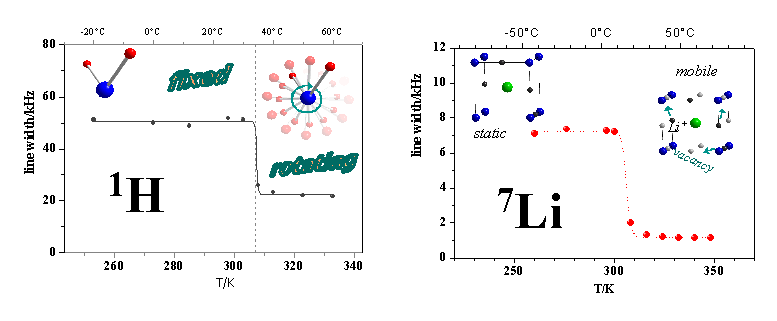
The ever increasing demand for high performance lightweight batteries for mobile microelectronic devices in health care and communication has triggered a wealth of studies on lithium solid state ionic conductors. Fundamental advantages over the current liquid electrolyte technology include increased shock resistance, thermal stability and environmental compatibility. Our main focus is the study of crystalline ionic conductors, in which the motion of the ion conducting species, predominantely Li+ or Na+, is supported by a rotation of the – often complex anions (see also). A variety of complementary characterization methods is used (powder diffraction, DSC, impedance spectroscopy, solid state NMR, ...) among which solid state NMR proves to be the most versatile and informative approach. The figure below shows the 7Li and 1H spectra of the anti- perovskite LiOH2Br. The transition from the non-conducting into the ion-conducting phase at T = 306K manifests itself in a drastic reduction of the line widths in the 1H and 7Li spectra, indicating a correlated motion of the anionic and the ion conducting species.
Host guest interactions in zeolite structures
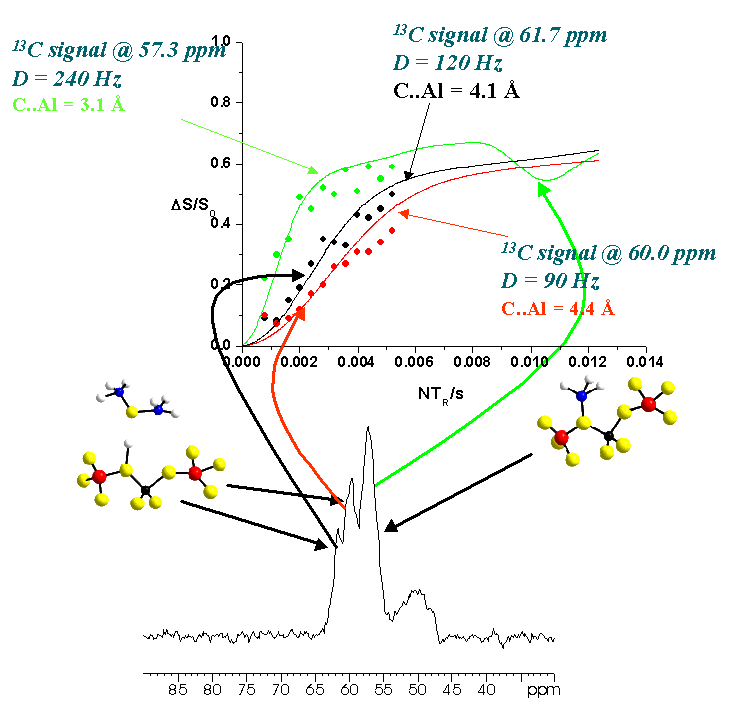
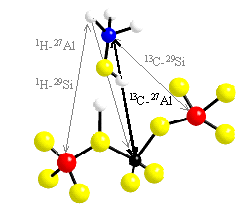
In these projects we study model systems to elucidate the reaction pathways of heterogenous catalysis of organic reactions using zeolite structures as solid acids. Even though the overall reaction and the influence of a given framework topology is more or less fairly understood, details of processes such as the MTG process (methanole to gasoline conversion) are currently under intense debate. Often discussed intermediates are a neutral adsorbate complex, an ionic adsorbate complex, a surface methoxy group and dimethyl ether. Our approach is to study model systems resembling the possible intermediates during the catalytic conversion. The figure illustrates the situation on the basis of a methanol molecule adsorbed to a zeolite fragment. A determination of internuclear distances allows for an accurate, model free description of the occuring adsorbate complexes. Looking at the natural abundances of the nmr active isotopes, any combination using 15N (guest) or 29Si (host) is not very promising. Using the 1H isotope (guest) has the disadvantage, that the dipolar couplings are often modulated by molecular reorienations (e.g. methyl group rotation). Furthermore, the spectral resolution of 1H is only very limited, leading to a superpostion of signals. The most promising combinations would be 13C (guest) and 27Al (host), a combination of nuclei, the study of which becomes possible using double resonance techniques developed in our group.
The following example (ZSM-5, loaded with methanole) illustrates, that the obtained distance information allows for unambiguous classification of the 13C signals. The signal at 57.3ppm obviously originates in carbon in surface methoxy groups (internuclear distance 3.1Å), the signals at 60.0ppm and 61.7ppm can be ascribed to carbon atoms in dimethyl ether.