Discovering a Highly Proton Conducting and Stable Oxide: The Case of Y-doped BaZrO3
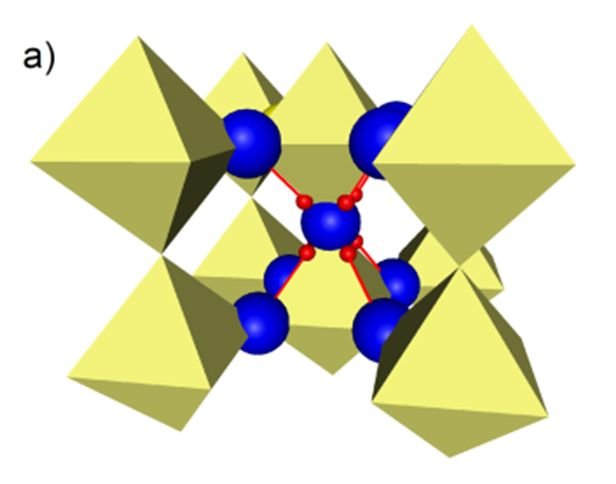 Y doped BaZrO3 with the cubic Perovskite structure shows a unique combination of high chemical stability, high water solubility and high proton conductivity. These properties are closely related to the high symmetry of its structure and the high entropy of protonic defects which are formed as a result of hydration.
Y doped BaZrO3 with the cubic Perovskite structure shows a unique combination of high chemical stability, high water solubility and high proton conductivity. These properties are closely related to the high symmetry of its structure and the high entropy of protonic defects which are formed as a result of hydration.
K. D. Kreuer
Aspects of the formation and mobility of protonic charge carriers and the stability of perovskite-type oxides
Solid State Ionics 125(1-4), 285–302 (1999).
DOI: 10.1016/S0167-2738(99)00188-5
K. D. Kreuer, St. Adams, W. Münch, A. Fuchs, U. Klock, and J. Maier
Proton conducting alkaline earth zirconates and titanates for high drain electrochemical applications
Solid State Ionics 145(1–4), 295–306 (2001).
DOI: 10.1016/S0167-2738(01)00953-5
K. D. Kreuer
Proton-Conducting Oxides
In Materials for Fuel Cells, K. D. Kreuer, ed., Vol. 33 of Annu. Rev. Mater. Res. 2003, Annual Reviews, Palo Alto, CA, 2003, pages 333–359.
DOI: 10.1146/annurev.matsci.33.022802.091825
K.-D. Kreuer
Mechanisms of Proton Conduction in Perovskite-Type Oxides
In Perovksite Oxide for Solid Oxide Fuel Cells, T. Ishihara, ed., Fuel Cells and Hydrogen Energy, Springer US, New York, USA, 2009, pages 261–272.
DOI: 10.1007/978-0-387-77708-5_13