Self-Assembly of Nanocrystalline Structures from Freestanding Oxide Membranes
The application of nanocrystals to human endeavors dates back to ancient Egyptian civilization. Until some 70 years ago, nanocrystals had been used extensively throughout history without the basic physics being understood and without characterization tools available to study them systematically. The emergence of nanoscience and nanotechnology has changed this fundamentally, meaning that we can now design nanomaterials. Nanoscience allows us to create nanocrystalline materials with novel properties that arise from quantum confinement and surface effects.
To synthesize such nanocrystals from functional materials, the team at MPI FKF has developed a new technique that utilizes self-assembly processes of complex oxides, driven by an inherent instability of thin films. This method of self-assembly-driven nanocrystal production yields highly oriented and excellently faceted nanocrystals down to 80 nm. Furthermore, nanowires and nanovoids with unprecedented orientation selectivity and crystalline purity up to the very edge of the nanostructure can be produced using this technique.
Traditionally, there have been two major categories of techniques to fabricate nanocrystals: top down and bottom up. A top-down technique means breaking down a bulk material to produce nanoscale structures, whereas a bottom-up technique synthesizes a nanostructure from scratch, using a variety of growth procedures. The authors have applied a combination of both approaches to create pristine nanocrystals. This was made possible by leveraging two simultaneous advances in oxide thin film growth achieved in the past five years. First, they used a selectively etchable, epitaxially grown buffer layer between the oxide thin film and the substrate, which can be etched away to release the thin film mechanically from the substrate in a membrane form. This enables the oxide membrane to be transferred in its freestanding form to any platform, where it can be processed further such that its properties can be altered. Second, they have applied in situ substrate heating capabilities developed in our own department. This technology uses CO2 laser irradiation for direct substrate heating, which allows the preparation of pristine substrate surfaces together with a capability to reach temperatures above 1000 oC quickly and cleanly inside a growth chamber.


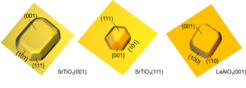
The starting point of the study was purely exploratory. The authors began by observing the large crystalline voids that appear upon annealing SrTiO3 membranes at 1000 °C on a prepared sapphire substrate. This prompted them to continue exploring the physics of dewetting, especially on membranes transferred on sapphire, as a sapphire surface is known to have a high diffusivity of adatoms. They found that, regardless of membrane thickness, the membranes went through the same stages of material agglomeration with increasing (albeit at different) temperatures. Furthermore, they investigated additional materials, all of which yielded a self-organized agglomeration, with the final stage always being nanocrystal formation.
This discovery provides a novel pathway for the clean nanostructuring of oxides, which has been historically challenging due to the oxides being rather chemically resistant to etching. Furthermore, top-down material removal approaches such as ion milling can damage the material at the nanostructure boundaries, thus altering the material properties where the nanostructure has been exposed to ion radiation. It also showcases the power of having the capability to alter the interface interaction via membrane liftoff and transfer, and it shows that freestanding membranes hold great promise for the future of nanoscience.
Harbola, Wu, Wang et al., Advanced Materials 2210989 (2023)
______________________________________________________________________________________________________________________